
Docking of Syk to FcεRI is enhanced by Lyn but limited in duration by SHIP1 | Molecular Biology of the Cell

Src Family Kinases Mediate Receptor-stimulated, Phosphoinositide 3-Kinase-dependent, Tyrosine Phosphorylation of Dual Adaptor for Phosphotyrosine and 3-Phosphoinositides-1 in Endothelial and B Cell Lines* - Journal of Biological Chemistry
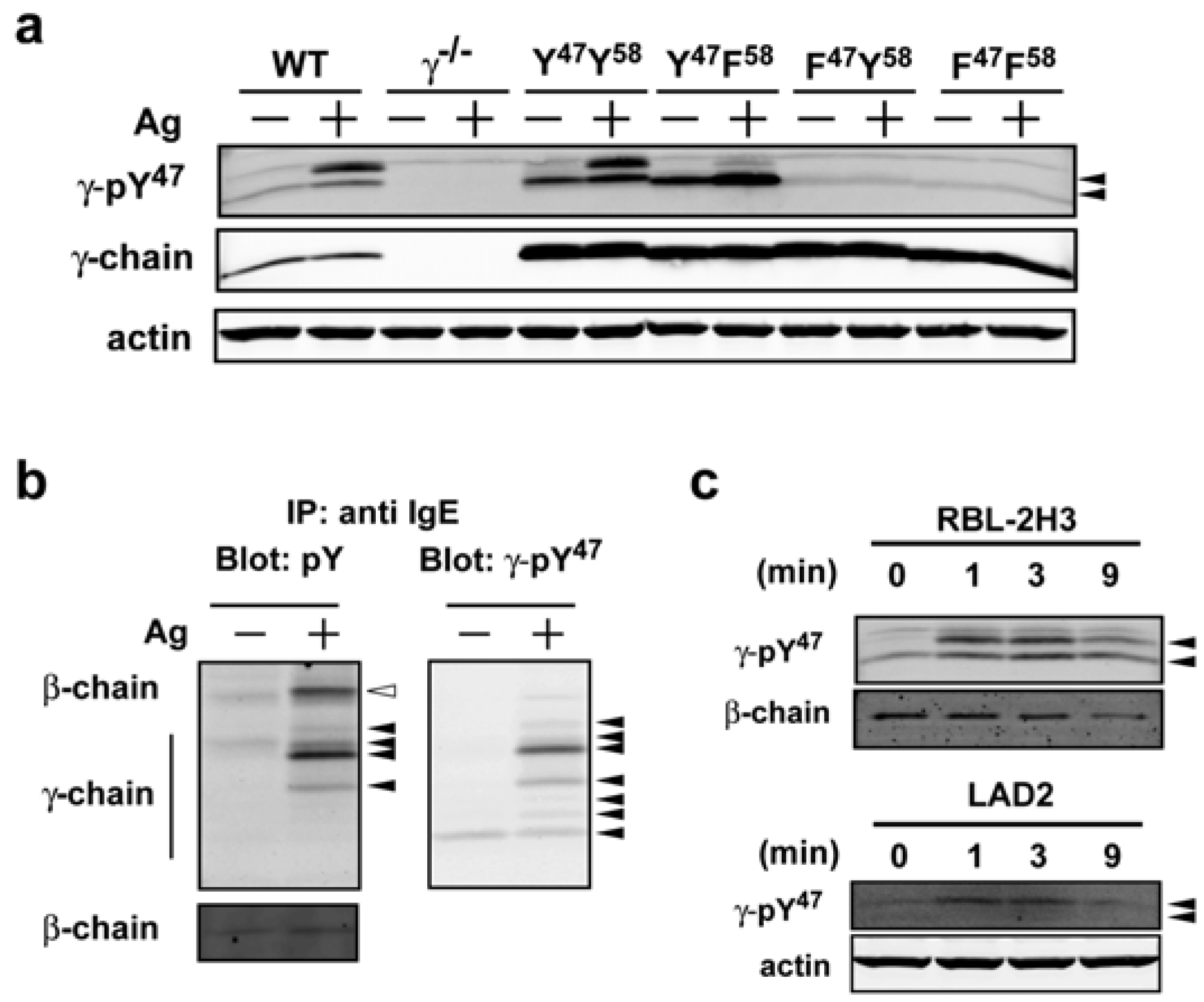
Antibodies | Free Full-Text | Characterization of a Phospho-Specific Antibody to the Fcε Receptor γ Chain, Reveals Differences in the Regulation of Syk and Akt Phosphorylation

Docking of Syk to FcεRI is enhanced by Lyn but limited in duration by SHIP1 | Molecular Biology of the Cell
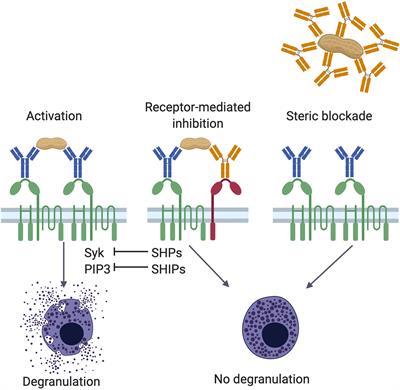
Frontiers | IgE and IgG Antibodies as Regulators of Mast Cell and Basophil Functions in Food Allergy

Ig-α-dependent translocation of SH2(SHIP1)-YFPv to the plasma membrane.... | Download Scientific Diagram

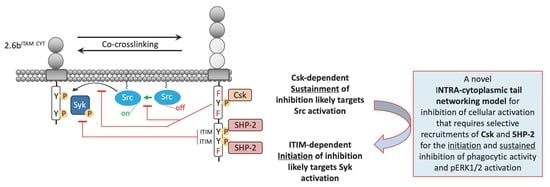
All-Atom Simulations Reveal the Intricacies of Signal Transduction upon Binding of the HLA-E Ligand to the Transmembrane Inhibitory CD94/NKG2A Receptor | Journal of Chemical Information and Modeling





![Comm - Sykla by Tetsushi -- Fur Affinity [dot] net Comm - Sykla by Tetsushi -- Fur Affinity [dot] net](https://d.furaffinity.net/art/tetsushi/1645752246/1645752240.tetsushi_sykla_comm_signed.png)

