![Calculate the number of molecules present in 0.5 moles of magnesium oxide ( MgO) . [Atomic weights : Mg = 24, O = 16 ] Calculate the number of molecules present in 0.5 moles of magnesium oxide ( MgO) . [Atomic weights : Mg = 24, O = 16 ]](https://dwes9vv9u0550.cloudfront.net/images/8090905/68318e51-f37d-486f-b3fb-f22fbc1043ce.jpg)
Calculate the number of molecules present in 0.5 moles of magnesium oxide ( MgO) . [Atomic weights : Mg = 24, O = 16 ]
![Calculate the number of molecules present in 0.5 moles of magnesium oxide ( MgO) . [Atomic weights : Mg = 24, O = 16 ] Calculate the number of molecules present in 0.5 moles of magnesium oxide ( MgO) . [Atomic weights : Mg = 24, O = 16 ]](https://dwes9vv9u0550.cloudfront.net/images/9085118/105391b5-2617-461a-9970-1a42367b289a.jpg)
Calculate the number of molecules present in 0.5 moles of magnesium oxide ( MgO) . [Atomic weights : Mg = 24, O = 16 ]

Question Video: Calculating the Mass of Oxygen Required to React with a Given Number of Magnesium Atoms | Nagwa
![Calculate the number of molecules present in 0.5 moles of magnesium oxide ( MgO) . [Atomic weights : Mg = 24, O = 16 ] Calculate the number of molecules present in 0.5 moles of magnesium oxide ( MgO) . [Atomic weights : Mg = 24, O = 16 ]](https://dwes9vv9u0550.cloudfront.net/images/9085118/c1781a1a-7e5f-47e8-a9bf-9b6d725abef3.jpg)
Calculate the number of molecules present in 0.5 moles of magnesium oxide ( MgO) . [Atomic weights : Mg = 24, O = 16 ]
A 3.250g sample of magnesium is burned in a container of 12.500g oxygen. What mass of oxygen gas remains unreacted after the magnesium has been completely consumed to form magnesium oxide as

OB: Intro to the Mole You gotta have a reference table and a calculator of your own now. No exceptions. - ppt download
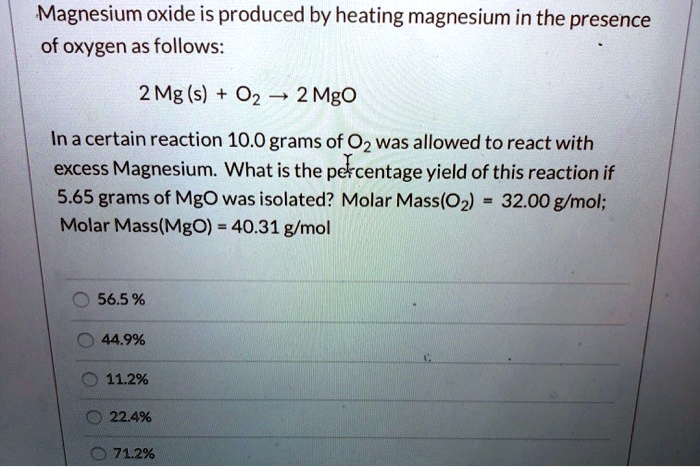
SOLVED: Magnesium oxide is produced by heating magnesium in the presence of oxygen as follows: 2 Mg (s) 02 2 Mgo In a certain reaction 10.0 grams of 0z was allowed to

1a.calculate the relative formula mass of magnesium oxide when the relative atomic masses are O=16 - Brainly.com

PDF) Experimentally Determining of the Empirical Formula of Magnesium Oxide | Folk Narongrit - Academia.edu