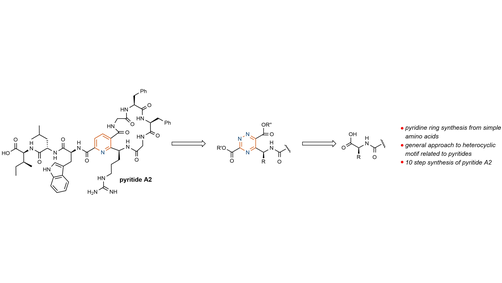
Angewandte Chemie on Twitter: "Bioinspired #TotalSynthesis of Pyritide A2 through Pyridine Ring Synthesis (Sarlah) @SarlahLab @ChemistryUIUC @IGBIllinois @UofIllinois #openaccess https://t.co/qZ6TrQjXjV https://t.co/e03eoGFXY9" / Twitter

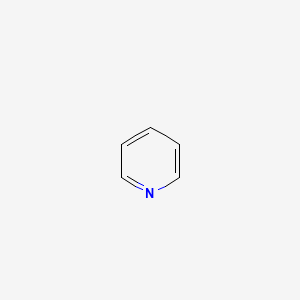
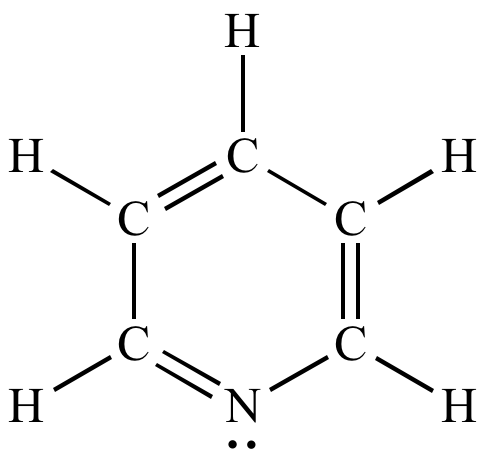
Pyridine is a benzene ring with one of the carbon atoms substituted with nitrogen. What are the hybridizations of atoms 1 and 2 respectively in the structure of pyridine? | Homework.Study.com
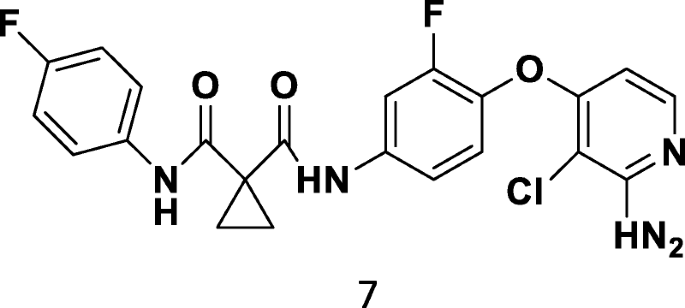
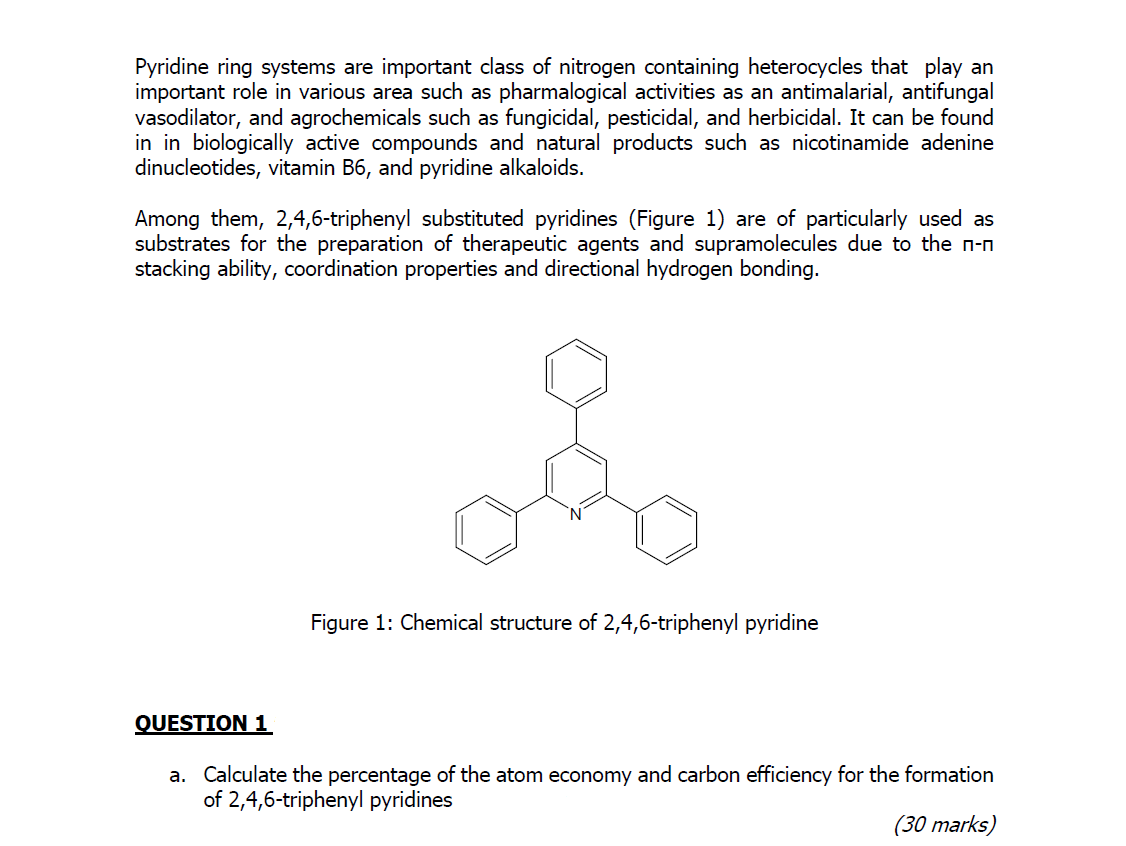
Medicinal attributes of pyridine scaffold as anticancer targeting agents | Future Journal of Pharmaceutical Sciences | Full Text
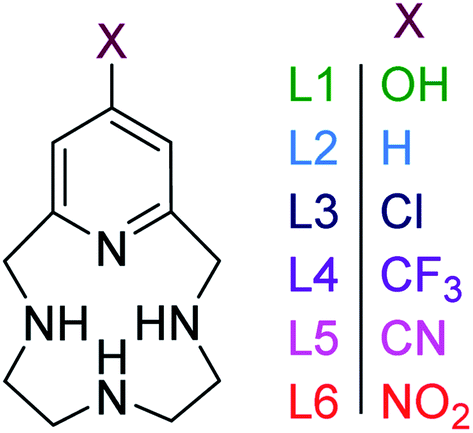
Functionalized pyridine in pyclen-based iron( iii ) complexes: evaluation of fundamental properties - RSC Advances (RSC Publishing) DOI:10.1039/D0RA05756H

Sequential Ring-Opening and Ring-Closing Reactions for Converting para-Substituted Pyridines into meta-Substituted Anilines | Organic Letters

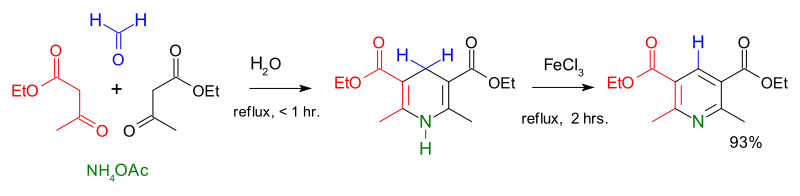
Synthesis of Nitrogen Heterocycles by the Ring Opening of Pyridinium Salts - Kearney - 2006 - Angewandte Chemie - Wiley Online Library

One-Pot Synthesis of Novel Functionalized Fused Pyridine Derivatives via Consecutive Pyrrolidine Ring-Closure/Ring-Opening/Formal Aza-Diels–Alder Reactions | The Journal of Organic Chemistry